Not all viruses are out to get us. How can we use some of their superpowers?
By Dr Luiza Wasiewska, Tyndall National Institute
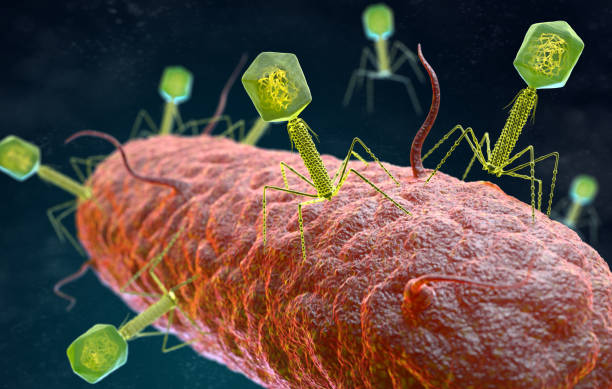
Hearing the word ‘virus’ gives many of us goosebumps, a reaction strongly influenced by the global COVID-19 pandemic, which killed millions of people and brought the world to a standstill for almost two years. However, not all viruses are harmful; in fact, some of them can even be beneficial to us. One example is bacteriophages, viruses that infect and kill bacteria. You may be surprised to learn that they are the most abundant organisms in the world, outnumbering bacteria by around ten to one! And if bacteria are everywhere, bacteriophages are everywhere too. They only prey on single-cell bacteria and are completely harmless to humans. Their complex relationships with bacterial communities have been studied in several research laboratories worldwide, and there is still so much more we can learn from them.
The growing global threat of antimicrobial resistance
One of the major global issues we are facing today is antimicrobial resistance (AMR). This occurs when bacteria that cause common infections and were once easily treated with antibiotics become resistant to them. As a result, bacterial infections that have been treatable since the discovery of antibiotics are becoming dangerous, and potentially lethal, once again. This has happened because antibiotics have not always been used correctly or with care, giving bacteria the chance to evolve and overcome them. Each year, approximately five million people worldwide die from infections by antimicrobial resistant-bacteria, and this number will continue to rise unless we act urgently.
Enter: bacteriophages – an old idea revisited
One promising application of bacteriophages is their use in fighting bacterial infections. Ironically, research in this area began more than a century ago, but it was largely neglected and forgotten in Europe and the US after the discovery of antibiotics. Today, the growing problems associated with the overuse of antibiotics have pushed researchers to revisit bacteriophages as a potential weapon in the fight against infection. However, this is not the only application where bacteria-eating viruses can be used.
Why faster detection matters
A crucial part of managing the AMR crisis is to improve bacterial detection and identification methods. If you ever had a sample taken to identify the cause of a urinary tract infection (UTI), you know that you often have to wait several days for the results. This is because our “gold standard” detection methods involve long and laborious processes. Faster methods, such as biosensors, do exist, but they must be proven to be extremely reliable before they can be widely implemented.
Biosensors are devices that allow the rapid detection of biological targets. One of the most well-known examples is the COVID-19 antigen test. Most of us have used these tests at least once since the onset of the COVID-19 pandemic. Biosensors usually contain two main components: one that recognises the target of interest (such as the COVID-19 virus), and another that converts that recognition into a measurable signal (such as a red line if the test is positive). These accessible, easy-to-interpret at-home tests have transformed how we manage infections.
Harnessing phage-derived molecules for faster, better biosensors
In my postdoctoral research, funded by Research Ireland, I am developing biosensors for the rapid detection of specific bacteria. The main innovation of my approach lies in how the target is recognised. Traditional biosensors, such as COVID or pregnancy tests, use antibodies to identify the target. Antibodies are proteins of the immune system that can recognise harmful invaders, but antibody extraction is expensive and requires the use of animals. This is where our useful viruses, the bacteriophages, may play an important role. To infect and kill bacteria, bacteriophages produce molecules called endolysins, which recognise and break down bacterial cell walls.
In Prof. Colin Hill’s laboratory in University College Cork (a collaborator on this project), these molecules are designed using the genetic information contained within bacteriophages, and then synthesised [6]. By attaching these molecules to our biosensor, we ensure that only the bacteria of interest will bind to it, allowing us to detect them. These molecules can be produced at scale, at low cost, and without the use of animals. I believe that this approach could revolutionise bacterial detection, making infection diagnosis possible in minutes rather than days – just like the COVID test.
Currently, we are focusing on the detection of Clostridioides difficile, more commonly known as C. diff., which is a major problem in hospitals. Although most of us carry this bacterium harmlessly in the gut, antibiotic treatment can cause an imbalance in the gut microbiome, allowing C. diff. to grow out of control, and cause real harm by producing dangerous toxins.
Looking forward
We aim to soon expand this sensing method to other pathogenic bacteria, with the long-term goal of detecting multiple problematic bacteria simultaneously. Detection in this project is done using electrochemistry, measuring small electrical signals produced by chemical reactions; the same principle behind well-known glucose sensors used for monitoring diabetes. Our sensors are fabricated using additive manufacturing, developed by my amazing mentor Dr. Sofia Teixeira, making them cheaper and minimising the need for specialist equipment. We are also exploring the use of more sustainable materials to make our sensors more environmentally friendly.
About the author:
Dr Luiza Wasiewska is a Research Ireland-funded Postdoctoral Fellow and Senior Researcher at the Tyndall National Institute. She combines her expertise in microbiology and biosensors to develop new diagnostic methods that could revolutionise how we detect and treat diseases. Her research focuses on alternatives to antibody recognition molecules which could be used not only to detect bacteria, but also other conditions, such as cancer, stress, or inflammation. In addition, she has a strong interest in sustainability and is researching the use of different sustainable materials (e.g. bioplastics) in the development electrochemical sensors.

Story image caption: Bacteriophages infecting a bacterium (image source: Shutterstock)